Wave-Particle Duality
Revisiting the Wave-particle Duality
Nowadays, physicists know that photons, particles, atoms, etc. propagate in the form of waves. However, the phenomenon of the transformation of waves into particles, and vice versa, is still obscure. This is the purpose of this chapter.
Historical Background of the Wave–Particle Duality
For centuries, the same question has been asked about light: is it composed of waves or particles?
- Waves: In 1690, Huygens proposed that light was a wave. Later, physicists such as Fresnel and Young supported this view. In 1873, Maxwell demonstrated that light is an electromagnetic wave.
- Particles: In 1704, Newton suggested that light was made of colored corpuscles, nowadays called 'photons'. This particle-like aspect was confirmed in 1905 by Einstein’s explanation of the photoelectric effect, which is used in solar panels.
We are still asking the same question today.
Is light a wave or a particle?
Current View of the Wave–Particle Duality
It is generally accepted that wave–particle duality represents two perspectives on the same phenomenon. Physicists often use a cylinder as an example to illustrate this puzzle: we observe either a rectangle or a circle, depending on the point of view.
Another version is the so-called "principle of superposition", a purely hypothet-ical idea with little real significance.
However, these theories do not explain what actually happens at the particle level, such that it sometimes behaves like a particle and sometimes like a wave.
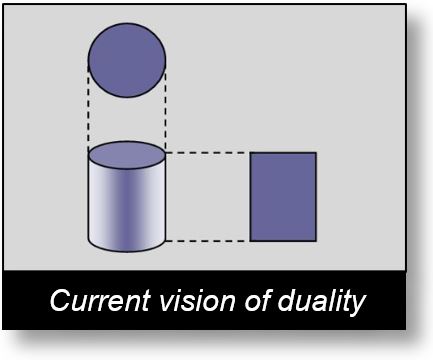
Understanding this Enigma
We have already provided the solution to the enigma of wave–particle duality in the introduction.
Take, for example, a body of water in a public garden. The water coming out of the filling pipe behaves like a particle, as shown in the photo below (white insert). Obviously, it is not a wave.
However, we observe that the water, which had a particle-like shape when leaving the pipe, transforms into waves in the basin (yellow insert).
This is precisely the definition of wave–particle duality.

Here, the water takes two forms: a particle-like state when leaving the pipe and a wave state in the basin. As demonstrated by physics experiments conducted over the past 100 years, the water in the basin behaves either as a particle or as a wave, but not both at the same time.

Examples of Wave-Corpuscle Duality
Other examples of wave–particle duality exist in nature, such as Niagara Falls. The water as it falls behaves like a particle. However, once it reaches the lower lake, the particle-like state of the water transforms into a wave-like state.
The reverse also exists. The water in a bathtub creates surface waves, which are like waves. But when the same water flows through the drain, it takes on a corpuscular nature.
The examples in the table below share a common feature:
- When the wave and the particle are of different natures, such as a stone and water, the wave-particle duality remains a genuine enigma.
- Conversely, when the wave and the particle pertain to the same substance, the enigma becomes much easier to explain.
Note: We did not discuss the medium, which is the environment in which the phenomenon occurs. This is because the wave and the medium are inseparable. Indeed, a wave necessarily propagates through a medium—in these examples, water. This topic will be addressed later.

Principles of Wave-Corpuscle Duality
To conclude our findings, it is useful to group the conclusions into three main principles that define the characteristics of wave–corpuscle duality. These principles have been confirmed by numerous experiments and are considered reliable — except in the case of photons, which will be discussed later in Part 4.
1. First Principle of Duality
This first principle is the most fundamental of all.
Duality can exist if, and only if:
- the wave,
- and the corpuscle,
are of the same nature.
2. Second Principle of Duality
As we have observed, when an object remains stationary, it exhibits a corpuscular nature. However, when it is in motion, it takes on a wave-like form. This principle is supported by electromagnetism: when electrons are immobile, such as when trapped in a capacitor, no electromagnetic activity is observed. But when electrons are in motion, they generate electromagnetic waves.
When the corpuscle moves,
it becomes a wave.
3. Third Principle of Duality
This fundamental principle is well known among physicists: the corpuscular state and the wave state cannot coexist simultaneously. In any given moment, matter expresses either its particle-like nature or its wave-like behavior—but never both at once. This mutual exclusivity lies at the heart of wave–particle duality and has been confirmed by countless experiments over the past century
A particle can only exist in one of the following states at any given moment:
- Corpuscle – when it is stationary,
- Wave – when in motion,
- Intermediate State – during the transition between rest and motion.
The Photon Exception
There is, however, an exception to the three main principles of wave–corpuscle duality: the photon. Although photons are considered corpuscles, they are not stationary. They only exist while traveling at the speed of 300,000 km/s. Theoretically, this would suggest they are waves rather than particles. The subject is more complex than it appears, because the photon, as a particle, does not truly exist. Physicists describe it as a “wave packet.” Despite its complexity, this enigma has a surprisingly rational explanation (see Part 4).
Overview of the Duality
Why has wave–particle duality been a mystery to physicists worldwide for more than a century?
In 1927 Davisson and Germer (Nobel Prize 1937) demonstrated electron diffraction, showing that electrons — like light — can exhibit wave-like behavior. Conversely, photons and other entities that behave like waves can also display particle-like properties. In other words, wave–particle duality is a single phenomenon manifested in particles, waves, and photons.
Possible explanation: The difficulty comes from the old, classical picture of electrons as tiny solid spheres orbiting the atomic nucleus. That picture is not correct (see Part 3). In fact, the electron behaves more like a cloud of charges than a tiny point particle, as it is often represented. That is probably why physicists find it so difficult to grasp duality.

The following pages will attempt to determine the constitution of matter — electrons, protons, etc. As you will see, the results may be surprising.
Perspectives of the Wave-Particle Duality
Nowadays, many physicists still envision elementary particles and photons as tiny solid spheres. This outdated view hinders progress and obstructs a deeper understanding of wave–particle duality and quantum mechanics. The solution to this puzzle will be revealed later in this part.
Summary on Wave-Corpuscle Duality
Wave–particle duality has long been seen as a burden by physicists, as no one has yet explained — using logic and common sense — how waves transform into particles and vice versa. However, the Spacetime Model views this duality not as a limitation, but as a powerful advantage. The reason is simple:
Wave–particle duality here explained already helps solve many of quantum mechanics’ deepest puzzles — such as the nature of quarks, antimatter, and electromagnetism. These topics will be explored further.
Even more compelling, duality offers insight into the internal structure of elementary particles. For instance: what is the true constitution of the electron? That’s what we’re now about to investigate.